Home > Teams > Metabolic and Cardiovascular Diseases > HCEMM- USZ Cerebral Blood Flow and Metabolism Research Group
Group Leader: Eszter Farkas Ph.D., D.Sc.
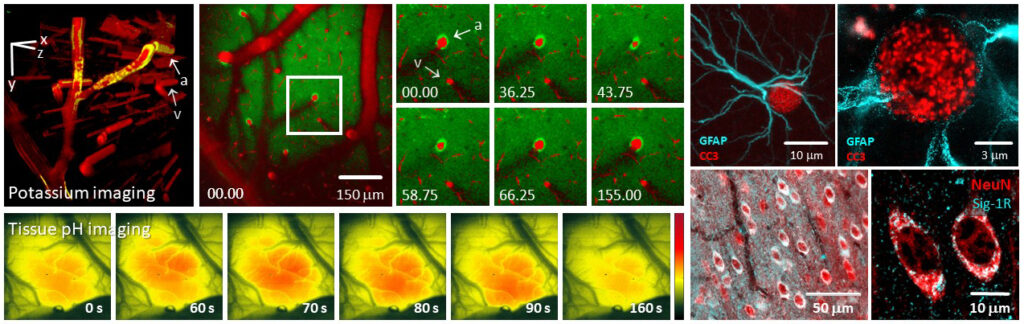
The evolution of cerebral edema is a life-threatening condition, yet our concept of how brain edema develops is incomplete, and treatment options remain limited. The current project addresses two challenges: (i) We will develop a new strategy for timely, accurate diagnosis to predict cerebral edema formation; and (ii) will offer novel, targeted, non-invasive and personalized therapy for the effective alleviation of brain edema formation. Furthermore, we posit that astrocyte swelling – as seen in a preliminary study of ours – and reactive astrogliosis typical of cerebral ischemia are directly and causally linked, and mediate edema-related neuronal injury. We will establish preclinical rodent models of cerebral ischemia and associated edema formation, and will monitor intracranial pressure with a telemetry based wireless system in freely moving animals for a clinically relevant time window. We will monitor astrocyte swelling at chosen time points with fluorescent astrocyte markers and multi-photon microscopy, and identify the reactive phenotype of astrocytes with immunolabeling and confocal microscopy. Led by our hypothesis that the serum level of endothelin 1 (ET-1) and natriuretic peptides – both produced by reactive astrocytes – are potential biomarkers to reflect cerebral edema formation, we will measure the blood concentration of ET-1 and natriuretic peptide repeatedly with ELISA, and match the data with intracranial pressure and the volume of the ultimate brain infarction. Because ET-1 is suggested to initiate astrogliosis by the activation of astrocytic endothelin B (ETB) receptors, we will explore this mechanism in conditional astrocytic ETB receptor knock-out ischemic mice. We will examine the potential of pharmacological ETB receptor antagonism, as a pertinent means to break the ET‑1/ETB receptor signaling cycle. This is then expected to suppress astrocyte reactivity, prevent the formation of cerebral edema, and ultimately, achieve neuroprotection.